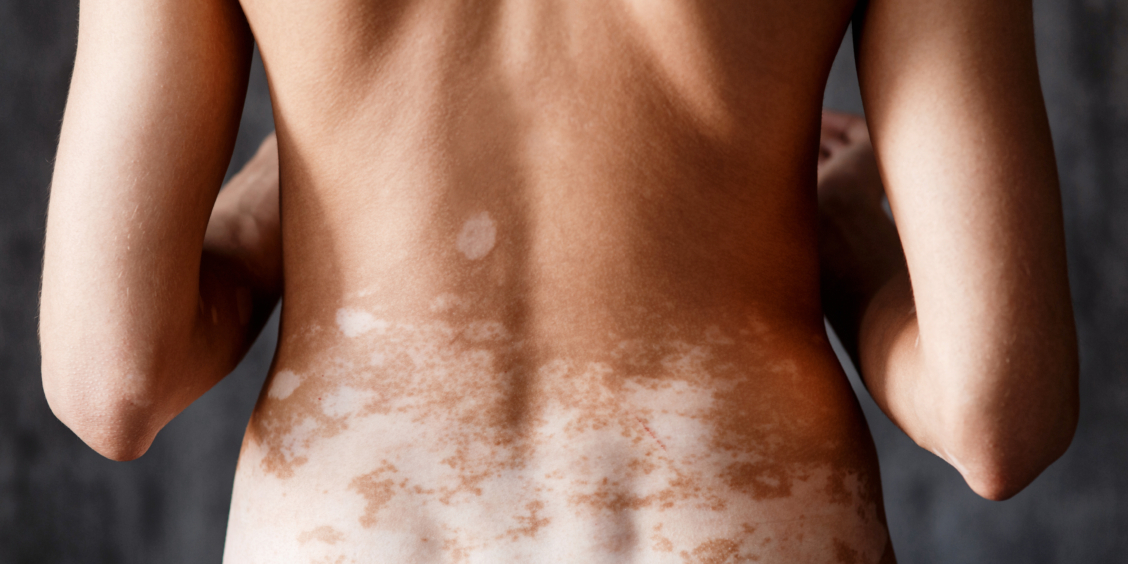
Today marks a groundbreaking moment for the vitiligo community: NICE has officially approved ruxolitinib (Opzelura) for NHS use in the treatment of non‑segmental vitiligo with facial involvement. This is the first treatment ever approved on the NHS specifically for repigmentation — a landmark moment that has the potential to transform lives.
This approval is not just a scientific milestone. It is a moment of recognition for everyone living with vitiligo. It says: your condition matters, your experiences matter, and your needs deserve to be met.
Today’s decision marks a historic turning point for people living with vitiligo. For decades, our community has waited for a licensed treatment to be recognised and funded by the NHS — and now, at last, that moment has arrived. This approval is not only a medical milestone; it is a recognition of every person who has felt unseen, unheard, or dismissed because of a visible difference. It tells our community that your experiences matter, your wellbeing matters, and your condition deserves real, evidence‑based care.
I want to extend my deepest gratitude to the 1,500 individuals who shared their personal stories, wrote to their MPs, and stood with us throughout this lengthy appraisal process. Your voices shifted the national conversation and were instrumental in achieving this outcome. While today is a moment to celebrate, it is also the beginning of a new chapter. We know people will have questions about access, eligibility, timelines and referrals. The Vitiligo Society will be here every step of the way to guide, support and advocate for you.
This is a victory made possible by community action — and a powerful reminder of what we can achieve when we stand together.
– Gurdeep Romanay, Chairperson, The Vitiligo Society
A Journey Defined by Determination, Community, and AdvocacyWhen NICE initially announced in 2025 that it would not recommend ruxolitinib for NHS use, the decision was felt deeply by our community. After decades without a licensed repigmentation treatment, this outcome was heartbreaking. But it also sparked something powerful.
Over 1,500 people engaged directly in our campaign — sharing their lived experiences, their struggles, and the emotional impact of visible difference. Many wrote to MPs, prompting Parliamentary questions and elevating vitiligo treatment access into the national conversation.
We met with NHS England and policy decision‑makers and submitted some of the largest volumes of patient evidence ever seen in a vitiligo appraisal. You trusted us with your stories, and we carried them into every conversation.
This collective effort was instrumental in ensuring that discussions between Incyte, NHS England and NICE continued long after initial rejection. Those conversations, powered by your voices, led directly to today’s historic approval.
What the Approval Means And What Happens NextUnder NHS implementation rules, newly approved medicines must be made available within 90 days of the final NICE decision. This means ruxolitinib will not be available immediately, but within approximately three months after the final guidance is published, NHS dermatology clinics will begin offering it. Key Practical Points:
Ruxolitinib will only be available through specialist dermatology services: This is a secondary care (hospital) treatment, not something GPs can prescribe. You must be under (or referred into) an NHS dermatology service.
It is approved specifically for non‑segmental vitiligo with facial involvement: This reflects the evidence NICE assessed and the terms of the medicine’s licence.
Access may vary between hospitals during the rollout: As with any new treatment, local pathways may take time to establish.
You may need a dermatology referral if you do not currently have one.
We know that this approval will raise practical questions, excitement, and understandable concerns — and we are prepared.
How Ruxolitinib Opens the Door for Future TreatmentsThis approval is not only a victory in its own right; it marks the beginning of a new era. In early 2026, systemic (whole‑body) treatments for vitiligo made major global progress. Upadacitinib (RINVOQ®), a once‑daily oral JAK inhibitor, was submitted to both the FDA and EMA in Feb 2026, as the first potential systemic pill for vitiligo after strong Phase 3 results.
Ruxolitinib’s approval demonstrates that vitiligo treatments can meet regulatory thresholds, paving the way for more innovations to follow. This is a shift many thought impossible a decade ago. Now it is happening.
A Moment of Gratitude for Every Voice RaisedWe want to thank every single person who:
Completed our calls for evidence Wrote to their MP Shared their personal story Engaged with our campaigns Joined our appeals Supported us as members Trusted us to represent themThis positive decision marks a significant milestone for people living with non-segmental vitiligo in the UK, and we are so pleased to be able to provide the first licensed treatment option to eligible patients.
This patient community has been without innovative treatment options for too long and this decision demonstrates the commitment of decision-makers to supporting and empowering people living with vitiligo to effectively manage their condition.
This decision is the result of years of close collaboration between The Vitiligo Society, the wider advocacy community and key healthcare system stakeholders to reach a positive outcome for patients, and we are so grateful for their determination and continued engagement.
On behalf of Incyte, we would like to thank all of our partners for their continued partnership to reach a positive outcome.
This approval belongs to you — the community who refused to quietly accept the message that vitiligo doesn’t matter. Today, we have proved that it does.
~ Pete Williams, General Manager, Incyte Biosciences UK
Questions? Need Guidance? We’re Here.As the implementation period begins, many people will naturally have questions:
Am I eligible? When will my hospital offer the treatment? What do I say to my dermatologist? What if my vitiligo is not on my face? What do I do if my GP isn’t aware of the treatment yet?You can contact us directly with any questions or concerns. Vitiligo Society Helpline: 0203 778 2204