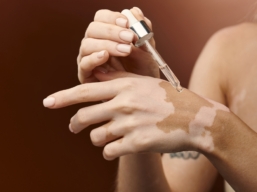
Ritlecitinib – a new vitiligo treatment?
Sorry, but you do not have permission to view this content.
Please support our work! You've enjoyed 1 article this month and we hope you have found it useful. Our work is entirely funded by memberships and donations, so please consider joining our charity today and supporting our work.
Become a Member
Already a member? Log in