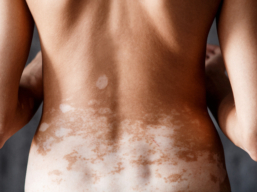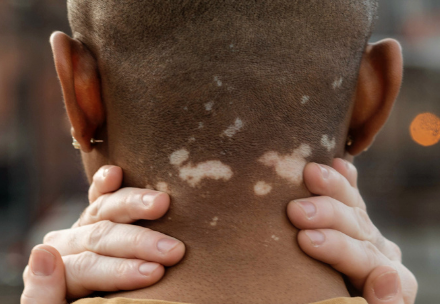
Final NICE Guidance on Ruxolitinib Published Today — A New Chapter for Vitiligo Treatment
Today marks another historic step forward for our community: NICE has published its final guidance (TA1140) recommending ruxolitinib (Opzelura) for non‑segmental vitiligo in people aged 12 and over. Read the full guidance HERE.
This follows the incredible momentum we built together earlier this year. On 24th February, we shared the groundbreaking news that NICE had overturned its earlier decision and approved ruxolitinib for NHS use, the first ever licensed treatment for vitiligo on the NHS. Your stories, signatures, lived experiences, and advocacy were central to this victory.
Today’s publication makes the decision official, final, and actionable across the NHS.
What the Final Guidance Says (Published Today)
NICE confirms that ruxolitinib cream is recommended for patients over the age of 12 with non‑segmental vitiligo. Ruxolitinib will only be offered on the NHS when first‑line topical treatments, such as topical corticosteroids or calcineurin inhibitors, have not worked or are not suitable. This is known as second‑line treatment.
The guidance does not apply to children under 12 or patients with segmental vitiligo. While clinicians may prescribe off‑label when they judge it clinically necessary, NICE emphasises that recommendations must be applied alongside individual clinical needs. Evidence of the effectiveness of ruxolitinib for treating for young children and segmental vitiligo remains limited. Some patients may wish to explore private access, where off‑label prescribing is possible, though this would require specialist agreement and would involve covering the full cost (with an indicative list price of £657 for a 100 g tube) in addition to the costs of private healthcare appointments.
What Happens Next Across the NHS?
Because this is a NICE Technology Appraisal:
- England is legally required to make the treatment available within the usual implementation window (90 days from publication), which applies to all NICE TAs.
- Wales automatically adopts all NICE Technology Appraisals under its Service Level Agreement with NICE, with a mandatory 60‑day funding requirement after publication.
As this guidance was published today (17 March 2026), NHS services will now begin the process of putting the treatment into place through dermatology specialists.
A New Era for Vitiligo Treatment
This final guidance is more than a technical decision — it is proof that meaningful change can happen when a community refuses to give up. Ruxolitinib’s approval opens the door to future treatments, including the first systemic (oral) therapies already progressing globally. The landscape is shifting in a way many believed impossible a decade ago.
Join Us to Shape the Next Chapter
The Vitiligo Society has been at the heart of every step forward — and the next breakthroughs will rely on the strength of our community.
Become a member today: https://vitiligosociety.org/membership
Your membership fuels advocacy, research influence, patient support, and the progress we are finally starting to see.
Please support our work! You've enjoyed 1 article this month and we hope you have found it useful. Our work is entirely funded by memberships and donations, so please consider joining our charity today and supporting our work.
Already a member? Log in